
CHEBI:28602
| Name | beta-D-fructofuranose 2,6-bisphosphate | 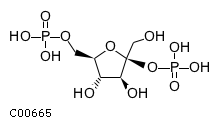 Download: mol | sdf |
| Synonyms | 02,6-di-o-phosphono--d-fructofuranose; Beta-d-fructose 2,6-bisphosphate; D-fructose 2,6-bisphosphate; Fdp; Fructose-2,6-diphosphate; Beta-d-fructofuranose 2,6-bisphosphate; | |
| Definition | A D-fructofuranose 2,6-bisphosphate with a beta-configuration at the anomeric centre. | |
| Molecular Weight (Exact mass) | 340.1157 (339.996) | |
| Molecular Formula | C6H14O12P2 | |
| SMILES | OC[C@]1(O[C@H](COP(O)(O)=O)[C@@H](O)[C@@H]1O)OP(O)(O)=O | |
| InChI | InChI=1S/C6H14O12P2/c7-2-6(18-20(13,14)15)5(9)4(8)3(17-6)1-16-19(10,11)12/h3-5,7-9H,1-2H2,(H2,10,11,12)(H2,13,14,15)/t3-,4-,5+,6+/m1/s1 | |
| InChI Key | YXWOAJXNVLXPMU-ZXXMMSQZSA-N | |
| Crosslinking annotations | KEGG:C00665 | 3DMET:B01302 | ChEBI:28602 | NIKKAJI:J470.690K | PDB-CCD:FDP | PubChem:3934 | |
| Pathway ID | Pathway Name | Pathway Description (KEGG) |
| map00051 | Fructose and mannose metabolism | NA |
| map04152 | AMPK signaling pathway | AMP-activated protein kinase (AMPK) is a serine threonine kinase that is highly conserved through evolution. AMPK system acts as a sensor of cellular energy status. It is activated by increases in the cellular AMP:ATP ratio caused by metabolic stresses that either interfere with ATP production (eg, deprivation for glucose or oxygen) or that accelerate ATP consumption (eg, muscle contraction). Several upstream kinases, including liver kinase B1 (LKB1), calcium/calmodulin kinase kinase-beta (CaMKK beta), and TGF-beta-activated kinase-1 (TAK-1), can activate AMPK by phosphorylating a threonine residue on its catalytic alpha-subunit. Once activated, AMPK leads to a concomitant inhibition of energy-consuming biosynthetic pathways, such as protein, fatty acid and glycogen synthesis, and activation of ATP-producing catabolic pathways, such as fatty acid oxidation and glycolysis. |
| map04922 | Glucagon signaling pathway | Glucagon is conventionally regarded as a counterregulatory hormone for insulin and plays a critical anti-hypoglycemic role by maintaining glucose homeostasis in both animals and humans. To increase blood glucose, glucagon promotes hepatic glucose output by increasing glycogenolysis and gluconeogenesis and by decreasing glycogenesis and glycolysis in a concerted fashion via multiple mechanisms. Glucagon also stimulates hepatic mitochondrial beta-oxidation to supply energy for glucose production. Glucagon performs its main effect via activation of adenylate cyclase. The adenylate-cyclase-derived cAMP activates protein kinase A (PKA), which then phosphorylates downstream targets, such as cAMP response element binding protein (CREB) and the bifunctional enzyme 6-phosphofructo-2-kinase/ fructose-2,6-bisphosphatase (one of the isoforms being PFK/FBPase 1, encoded by PFKFB1). |
| map05230 | Central carbon metabolism in cancer | Malignant transformation of cells requires specific adaptations of cellular metabolism to support growth and survival. In the early twentieth century, Otto Warburg established that there are fundamental differences in the central metabolic pathways operating in malignant tissue. He showed that cancer cells consume a large amount of glucose, maintain high rate of glycolysis and convert a majority of glucose into lactic acid even under normal oxygen concentrations (Warburg's Effects). More recently, it has been recognized that the 'Warburg effect' encompasses a similarly increased utilization of glutamine. From the intermediate molecules provided by enhanced glycolysis and glutaminolysis, cancer cells synthesize most of the macromolecules required for the duplication of their biomass and genome. These cancer-specific alterations represent a major consequence of genetic mutations and the ensuing changes of signalling pathways in cancer cells. Three transcription factors, c-MYC, HIF-1 and p53, are key regulators and coordinate regulation of cancer metabolism in different ways, and many other oncogenes and tumor suppressor genes cluster along the signaling pathways that regulate c-MYC, HIF-1 and p53. |

