
CHEBI:17985
| Name | adenosine 3',5'-bismonophosphate | 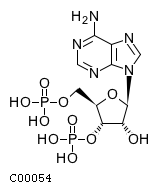 Download: mol | sdf |
| Synonyms | 03'-phosphoadenosine 5'-phosphate; 3'-phosphoadenylate; Adenosine 3',5'-bisphosphate; Na; Pap; Phosphoadenosine phosphate; | |
| Definition | An adenosine bisphosphate having two monophosphate groups at the 3'- and 5'-positions. | |
| Molecular Weight (Exact mass) | 427.2011 (427.0294) | |
| Molecular Formula | C10H15N5O10P2 | |
| SMILES | Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(O)=O)[C@@H](OP(O)(O)=O)[C@H]1O | |
| InChI | InChI=1S/C10H15N5O10P2/c11-8-5-9(13-2-12-8)15(3-14-5)10-6(16)7(25-27(20,21)22)4(24-10)1-23-26(17,18)19/h2-4,6-7,10,16H,1H2,(H2,11,12,13)(H2,17,18,19)(H2,20,21,22)/t4-,6-,7-,10-/m1/s1 | |
| InChI Key | WHTCPDAXWFLDIH-KQYNXXCUSA-N | |
| Crosslinking annotations | KEGG:C00054 | 3DMET:B01140 | CAS:1053-73-2 | ChEBI:17985 | ChEMBL:CHEMBL574817 | KNApSAcK:C00019352 | NIKKAJI:J14.396K | PDB-CCD:A3P | PubChem:3356 | |
| Pathway ID | Pathway Name | Pathway Description (KEGG) |
| map00230 | Purine metabolism | NA |
| map00532 | Glycosaminoglycan biosynthesis - chondroitin sulfate / dermatan sulfate | Glycosaminoglycans (GAGs) are linear polysaccharide chains consisting of repeating disaccharide units and form proteglycans by covalently attaching to their core proteins. Chondroitin sulfate (CS) is a glycosaminoglycan with the disaccharide unit of beta-D-galactosamine (GalNAc) and beta-D-glucuronic acid (GlcA), and often modified with ester-linked sulfate at certain positions. Dermatan sulfate (DS) is a modified form of CS, in which a portion of D-glucuronate residues is epimerized to L-iduronates (IdoA). CS and DS are linked to serine residues in core proteins via a linkage tetrasaccharide formed by the transfer of xylose and three more residues [MD:M00057]. The assembly process of CS is initiated by transferring GalNAc residue to the linkage tetrasaccharide. The polymerization is catalyzed by bifunctional enzymes (chondroitin synthases) possessing both beta 1,3 glucuronosyltransferase and beta 1,4 N-acetylgalactosaminyltransferase activities [MD:M00058]. Chondroitin polymerization also requires the action of the chondroitin polymerizing factor. There are various O-sulfation patterns in CS and DS, where 4-O sulfation and 6-O sulfation of GalNAc and 2-O-sulfation of the uronic acids (GlcA / IdoA) are mainly found. |
| map00534 | Glycosaminoglycan biosynthesis - heparan sulfate / heparin | Heparan sulfate (HS) and heparin (Hep) are glycosaminoglycans with repeating disaccharide units that consist of alternating residues of alpha-D-glucosamine (GlcN) and uronic acid, the latter being either beta-D-glucuronic acid (GlcA) or alpha-L-iduronic acid (IdoA). In these sugar residues, sulfation modification may be performed at various positions. Structural studies show that Hep possesses a higher degree of sulfation than HS. The biosynthesis of HS/Hep occurs with the addition of the first GlcNAc residue by EXTL3 glycosyltransferase after completion of tetrasaccharide linkage region attached to serine residue of a core protein. The chain polymeraization is then catalyzed by EXT1 and EXT2 transferases. As the chain polymerizes, HS/Hep undergoes a series of modification reactions including N-deacetylation, N-sulfation, epimerization, and subsequently O-sulfation. As final products of biosynthesis, HS is present in the form of hepran sulfate proteoglycan (HSPG) whereas Hep exists as a sugar chain without a core protein. The proteoglycan families with HS, as well as CS (chondroitin sulfate), DS (dermatan sulfate), and KS (keratan sulfate), are composed of two main types depending on the subcellular locations: cell membrane and extracellular matrix [BR:ko00535]. HS/Hep has been shown to bind to a variety of molecules, such as growth factors, chemokines, morphogens, and extracellular matrix components [BR:ko00536]. |
| map00770 | Pantothenate and CoA biosynthesis | NA |
| map00920 | Sulfur metabolism | Sulfur is an essential element for life and the metabolism of organic sulfur compounds plays an important role in the global sulfur cycle. Sulfur occurs in various oxidation states ranging from +6 in sulfate to -2 in sulfide (H2S). Sulfate reduction can occur in both an energy consuming assimilatory pathway and an energy producing dissimilatory pathway. The assimilatory pathway, which is found in a wide range of organisms, produces reduced sulfur compounds for the biosynthesis of S-containing amino acids and does not lead to direct excretion of sulfide. In the dissimilatory pathway, which is restricted to obligatory anaerobic bacterial and archaeal lineages, sulfate (or sulfur) is the terminal electron acceptor of the respiratory chain producing large quantities of inorganic sulfide. Both pathways start from the activation of sulfate by reaction with ATP to form adenylyl sulfate (APS). In the assimilatory pathway [MD:M00176] APS is converted to 3'-phosphoadenylyl sulfate (PAPS) and then reduced to sulfite, and sulfite is further reduced to sulfide by the assimilatory sulfite reductase. In the dissimilatory pathway [MD:M00596] APS is directly reduced to sulfite, and sulfite is further reduced to sulfide by the dissimilatory sulfite reductase. The capacity for oxidation of sulfur is quite widespread among bacteria and archaea, comprising phototrophs and chemolithoautotrophs. The SOX (sulfur-oxidation) system [MD:M00595] is a well-known sulfur oxidation pathway and is found in both photosynthetic and non-photosynthetic sulfur-oxidizing bacteria. Green sulfur bacteria and purple sulfur bacteria carry out anoxygenic photosynthesis with reduced sulfur compounds such as sulfide and elemental sulfur, as well as thiosulfate (in some species with the SOX system), as the electron donor for photoautotrophic growth. In some chemolithoautotrophic sulfur oxidizers (such as Thiobacillus denitrificans), it has been suggested that dissimilatory sulfur reduction enzymes operate in the reverse direction, forming a sulfur oxidation pathway from sulfite to APS and then to sulfate. |

